We conduct research in medical imaging and image guidance. Our goal is to develop image guidance technologies based on functional measurements to guide interventional procedures. In order to meet these goals, we develop novel ultrasound transducers, contrast agents, systems, and image formation approaches. We then evaluate the performance of these technologies in laboratory experiments and in pre-clinical imaging studies.
Research directions
The technologies developed in our lab target address some of the following challenges.
Development of functional interventional imaging systems.
Despite the increasing utilization of minimally-invasive procedures over open surgery in the past 2-3 decades, many interventional procedures are still performed without any ability to visualize local anatomy or instrument position, let alone functional information such as hemodynamics, physiological motion, or tissue stiffness. In these “blind” procedures without any imaging, collaborating physicians rely on the “feel” of instruments as they manually navigate instruments through vasculature and interact with tissue. Furthermore, many interventionalists describe “forming a mental image of the situation” based on pre-procedural imaging and on the physical sensation communicated through touch during tissue-instrument interactions. Mental images and relying on touch are not the best approach for rapid, safe procedures. At best, current interventional imaging technology such as fluoroscopy allows guidance with 2D planar projection images that average complex 3D anatomy and use ionizing radiation. Alternatively, our lab develops systems for integrating the imaging system into the procedure to provide these missing imaging capabilities. For example, our work in 3D functional characterization of intermediate stenosis in coronary artery disease is funded by NIH. This work has been published here and here.
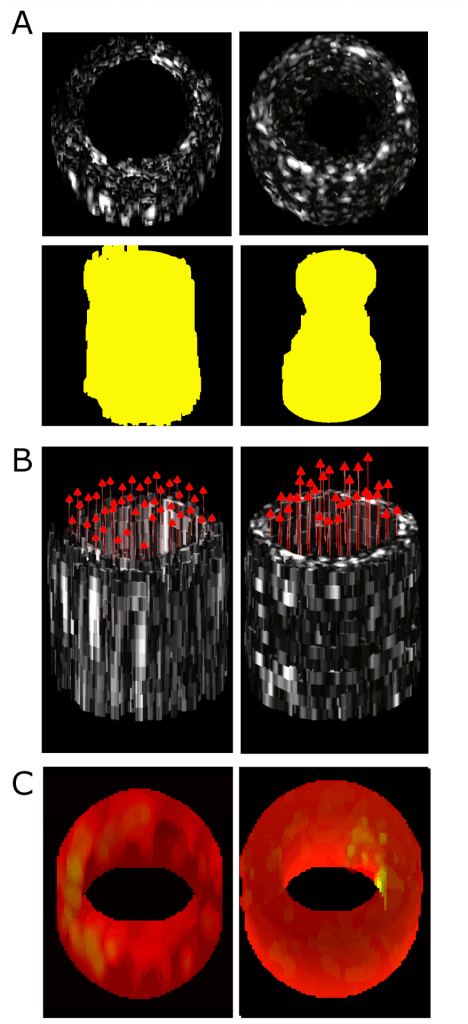
Development of integrated image-guided robotic systems.
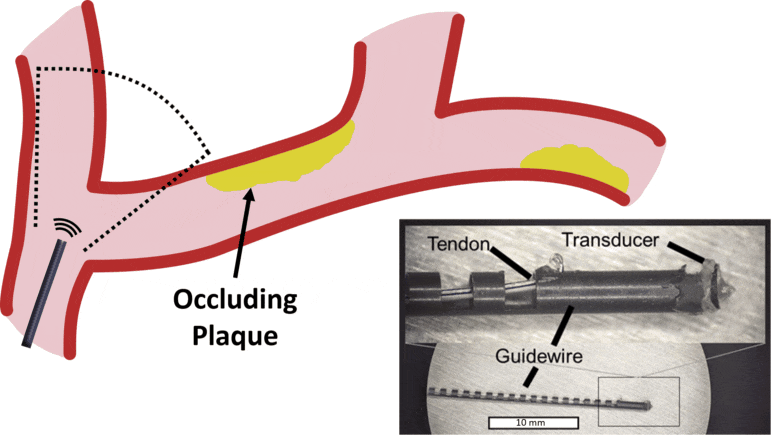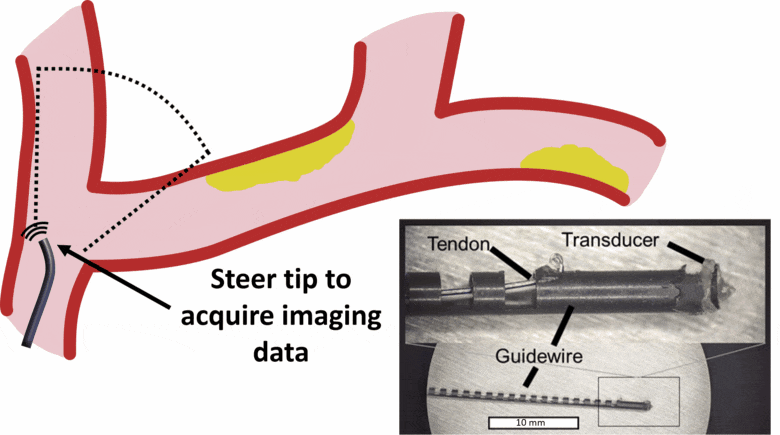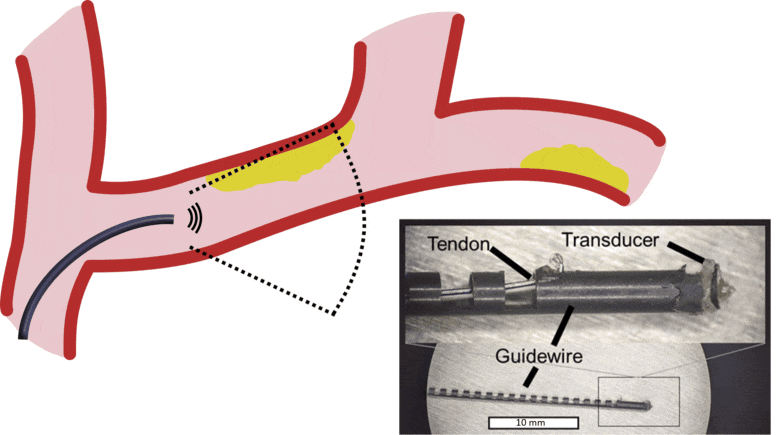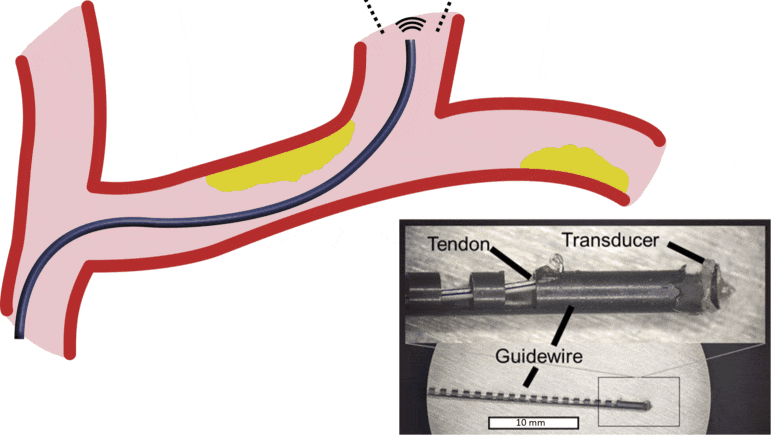
Separately from developing the actual imaging technology, an additional critical limitation in interventional procedures is the inability to safely guide instruments past obstacles in small anatomical spaces, such as arteries with heavily-calcified chronic occlusions or tortuous anatomy.
Our lab is developing new systems that integrate the instrument (e.g. steerable guidewire) with the imaging system. Development of a robotically-steered guidewire with integrated ultrasound imaging to overcome heavily calcified chronic total occlusions in peripheral artery disease is funded by an NIH R01 in collaboration with Jaydev Desai (BME), Zach Bercu (Interventional Radiology), and Khusrow Niazi (Interventional Cardiology). The first prototype system was published in the July 2021 issue of IEEE Transactions on Biomedical Engineering.
Development of image-guided therapy.
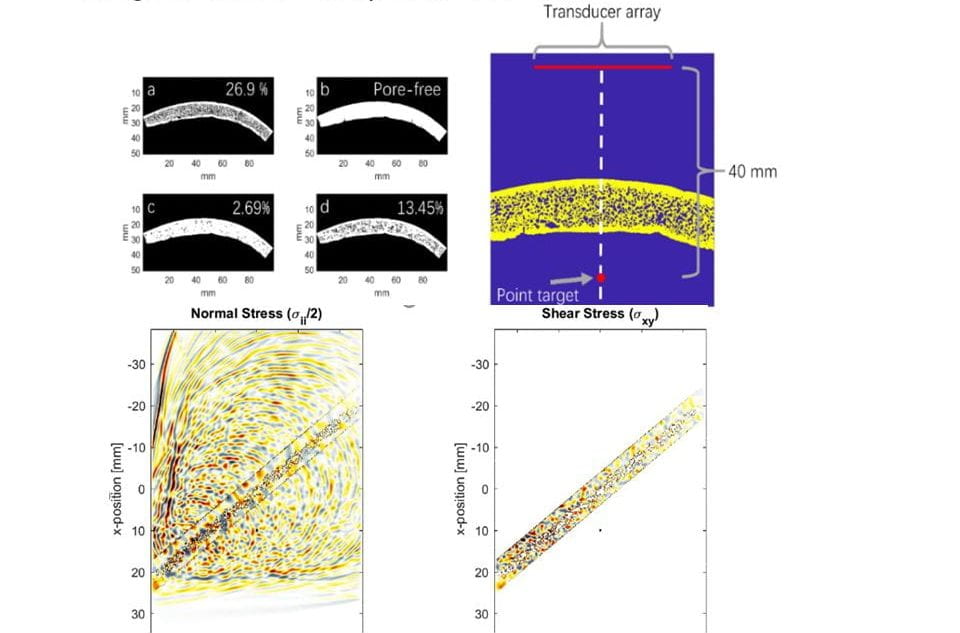
Both minimally-invasive and non-invasive therapy must safely deliver energy to treatment locations while sparing healthy tissues. The goal is to develop technologies for therapeutic guidance and monitoring to overcome challenges such as the skull, challenging anatomy (i.e. calcifications), and physiological motion. Systems and techniques for transcranial ultrasound imaging and monitoring of focused ultrasound therapy are developed in collaboration with Costas Arvanitis (BME/ME), Alper Erturk (ME), and Massimo Ruzzene (ME, University of Colorado) with funding from the National Science Foundation. This project investigates fundamentals of vibration and acoustic propagation in the skull-brain system. Specifically, our lab is investigating effects of the skull microstructure on imaging.